The value of compression factor at the critical state of a vander
The value of compression factor at the critical state of a vander waals gas is

Compressibility factor, Z of a gas is given as Z= frac { pV }{ nRT } (i) What is the value of Z an ideal gas?(ii) For real gas what will be
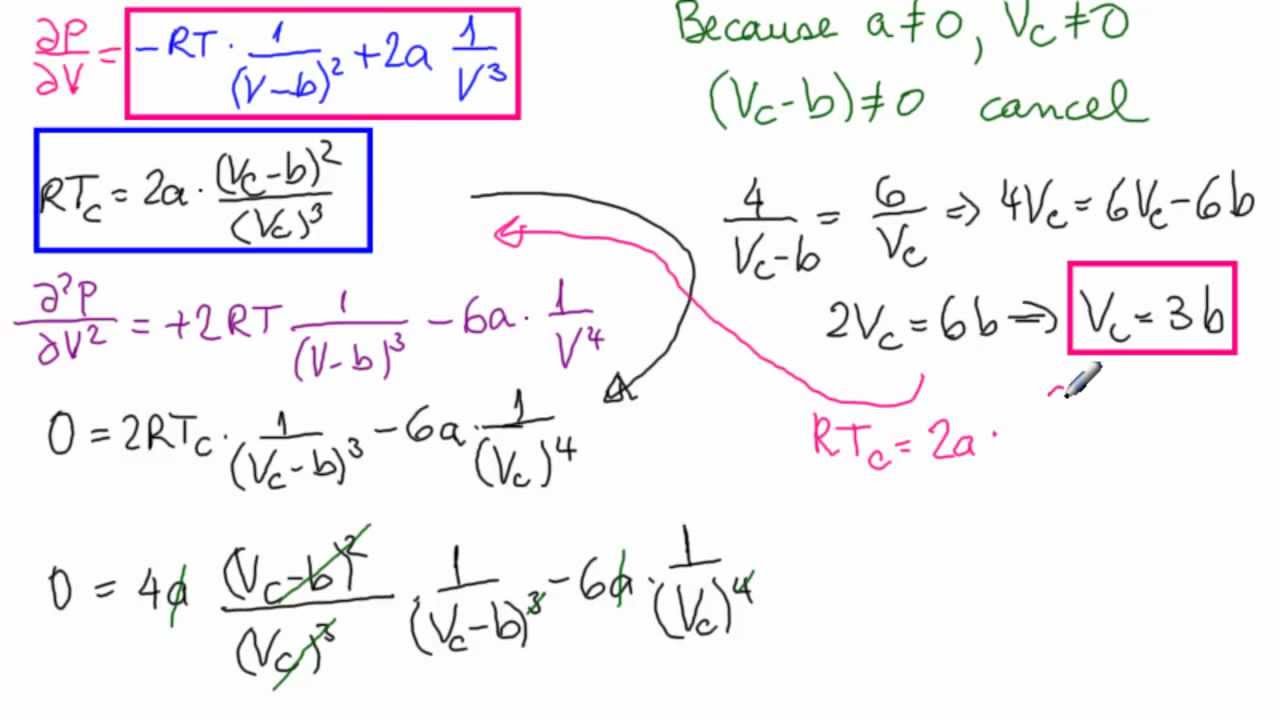
Critical Point with Van der Waal's Equation

The compressibility factor in terms of Pc, Vc and Tc is called Zc. Th

Solved Problem 1. Let's consider the van der Waals equation
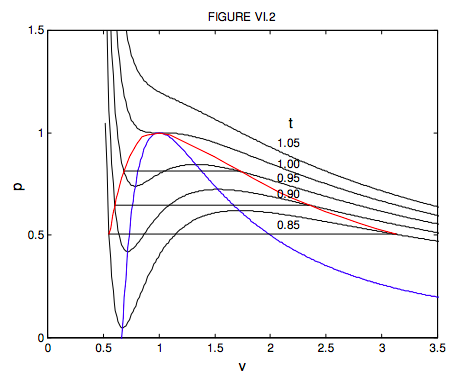
6.3: Van der Waals and Other Gases - Physics LibreTexts

The value of compressibility factor at the critical state the gas matc

Compressibility factor (gases) - Citizendium
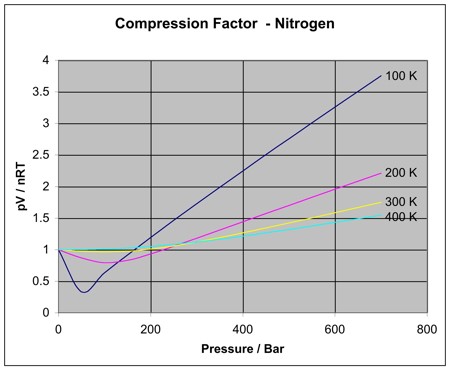
2.7: Real Gases - Chemistry LibreTexts

7.) Equation of state a gas is Where B and C are constant. the

Van der Waals Equation - Derivation, Relation Between Ideal Gas Law, Application

Compressibility factor - Wikipedia

Gaseous State.pdf - Chemistry - Notes - Teachmint
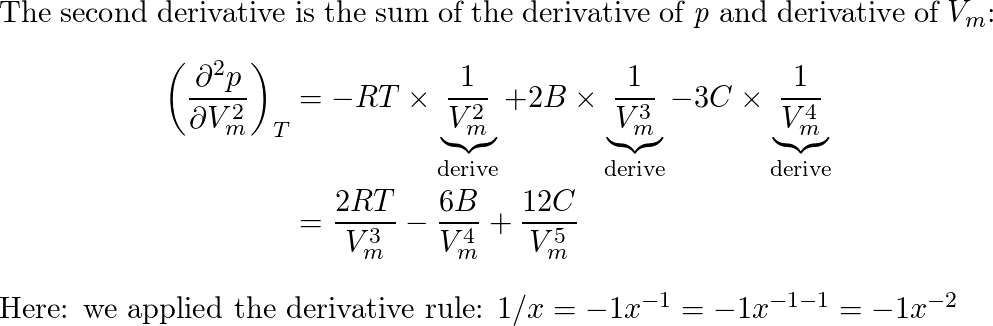
A scientist proposed the following equation of state $p= ra
What is the value of compressibility factor in terms of vander waal cons†an t at different conditions of pressure and volume?Why is Z>1 for H2 and He gas