Description of real gases: Compression factor

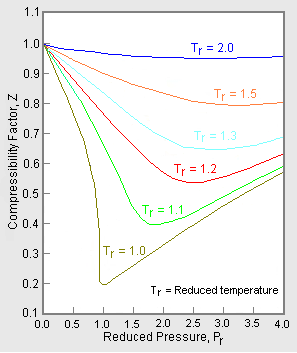
Compressibility factor (gases) - Citizendium
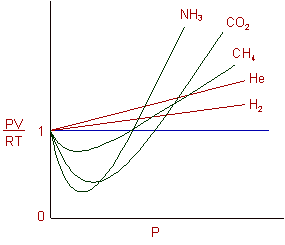
REAL GASES, DEVIATION FROM IDEAL GAS BEHAVIOUR
Ideal gases and real gases are compressible or not compressible

Compressibility factor, Z of a gas is given as Z= frac { pV }{ nRT
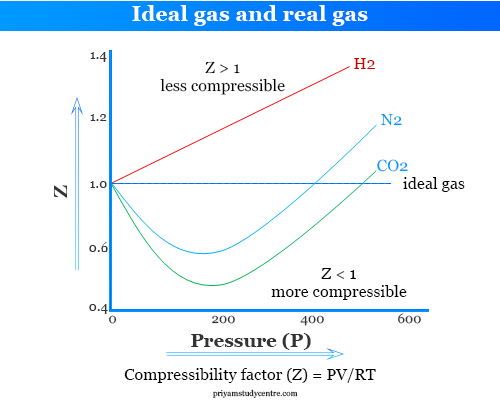
Ideal and Real Gases - Definition, Comparison, Properties
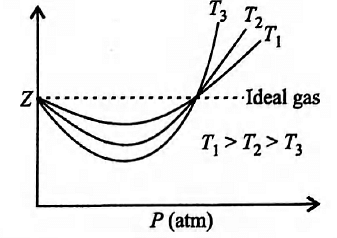
Boyle?? temperature or Boyle point is the temperat

Compressibility Factor - an overview

Non-Ideal Gas Behavior Chemistry: Atoms First
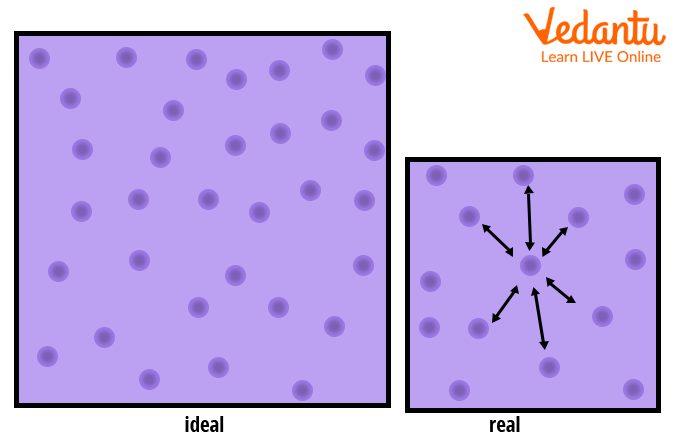
JEE - Compressibility Factor Important Concepts and Tips

Solved Real gas effects can be expressed as departures from

Behaviour of Real Gases, PDF, Gases