physical chemistry - Why do some gases have lower value of Z for a

In the above graph,the minima of the curve for methane is more than that of nitrogen. Also, for a given value of pressure, the value of $Z$ for methane is less than that of nitrogen. They seem to m

Lesson Explainer: Ionization Energy

What are the Properties of Gases? - Physical Properties Of Gases
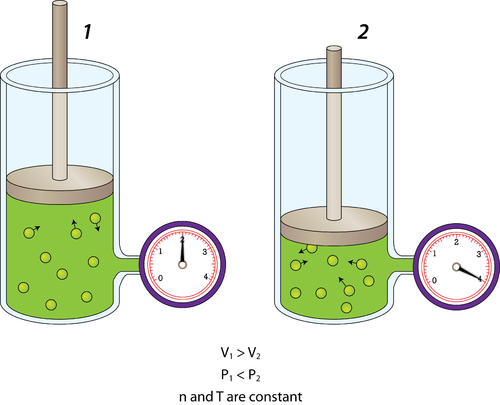
The Behavior of Gases Chemistry for Non-Majors

Gas Compressibility - an overview

Real Gas Behavior The Compression Factor (Z) [Example #2]
Gases, Free Full-Text

Deviation Of Real Gas From Ideal Gas Behavior

molar gas volume Avogadro's Law moles and mass calculations gcse chemistry calculations igcse KS4 science A level GCE AS A2 O Level practice questions exercises

Non-Ideal Gas Behavior Chemistry: Atoms First