At a high pressure, the compressibility factor (Z) of a real gas is us

At high P. P gt gt (n^(2)a)/(V^(2)) So ‘a’ can be neglected.

Compressibility factor (gases) - Citizendium

Non-ideal behavior of gases (article)
Van der waals equation: Derivation, Explanation

Gas Z Factor Calculator: Dranchuk-Abou-Kassem · PVT Solver
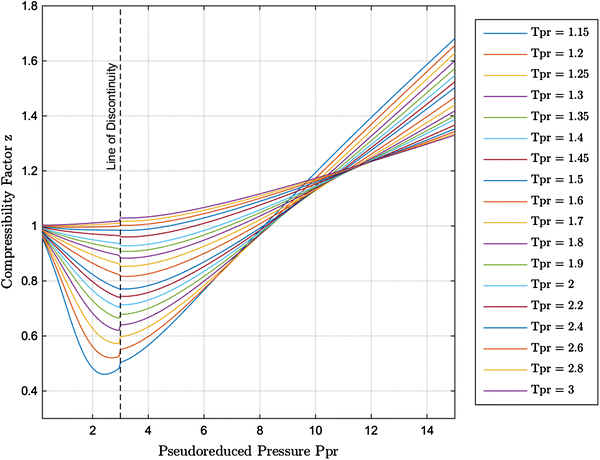
New explicit correlation for the compressibility factor of natural gas: linearized z-factor isotherms

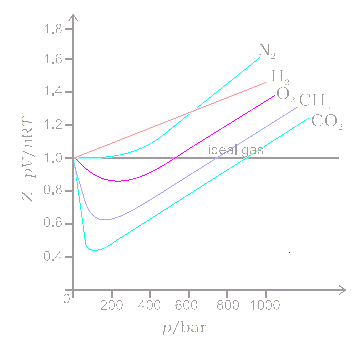
The given graph represents the variation of Z (compressibility factor) vs. P three real gases A, B and C. Identify the correct statementFor the gas A, a=0 and its dependence on P

3.3: Real gas and compressibility factor - Engineering LibreTexts

Compressibility Chart - an overview

Explain how the compression factor varies with pressure and

At a high pressure, the compressibility factor (Z) of a real gas is usual..

The density of air is 0.001293 gm/ml at S.T.P. It's vapour density is

PDF] Natural Gas Compressibility Factor Measurement and Evaluation for High Pressure High Temperature Gas Reservoirs
How does real gas occupies more volume than an ideal gas at high pressure? - Quora

COMPRESSIBILITY FACTOR

Solved The graph of compressibility factor (Z)v/sP for 1 mol