Compressibility factor Z = PV / nRT is plotted against pressure as shown below:What is the correct order for the liquefiability of the gases shown in the above graph? A. CO 2

Compressibility factor Z = PV / nRT is plotted against pressure as shown below:What is the correct order for the liquefiability of the gases shown in the above graph? A. CO 2
Compressibility factor Z - PV - nRT is plotted against pressure as shown below-What is the correct order for the liquefiability of the gases shown in the above graph- A- CO 2- CH 4- N 2- H 2B- H 2- CH 4- N 2- CO 2C- CH 4- H 2- N 2- CO 2D- H 2- N 2- CH 4- CO 2
Why does gas liquefy at high pressure? Even at high-pressure
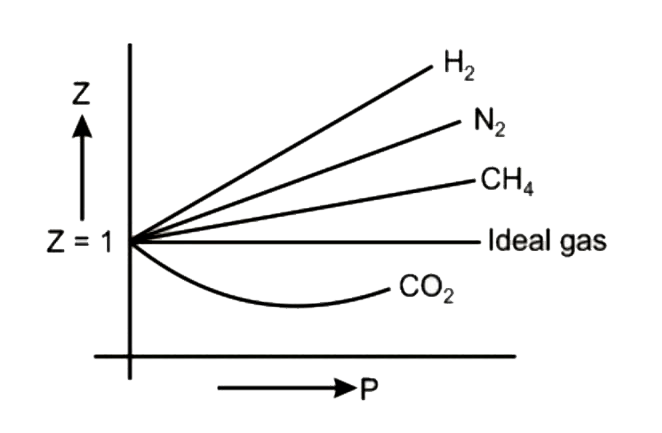
Compressibility factor (Z=(PV)/(nRT)) is plotted against pressure

Compressibility factor (Z=(PV)/(nRT)) is plotted against pressure
What is the significance of the curve part in Z vs. P graph of compressibility of a gas? - Quora

PDF) Field Operational Problems due to Condensate Formation in Retrograde Gas Reservoirs

Gas compressibility factor Z: Ideal gas vs Real gas

Compressibility Factor of Gas Overview, Equation & Chart
1642646504_391987.png

gaseous state

Multi-scale simulation of wave propagation and liquefaction in a one-dimensional soil column: hybrid DEM and finite-difference procedure

Sustainability June-2 2023 - Browse Articles