ISPE GAMP® 5: A Risk-Based Approach to Compliant GxP Computerized


ISPE GAMP® 5: A Risk-Based Approach to Compliant GxP Computerized Systems [Chinese Translation]
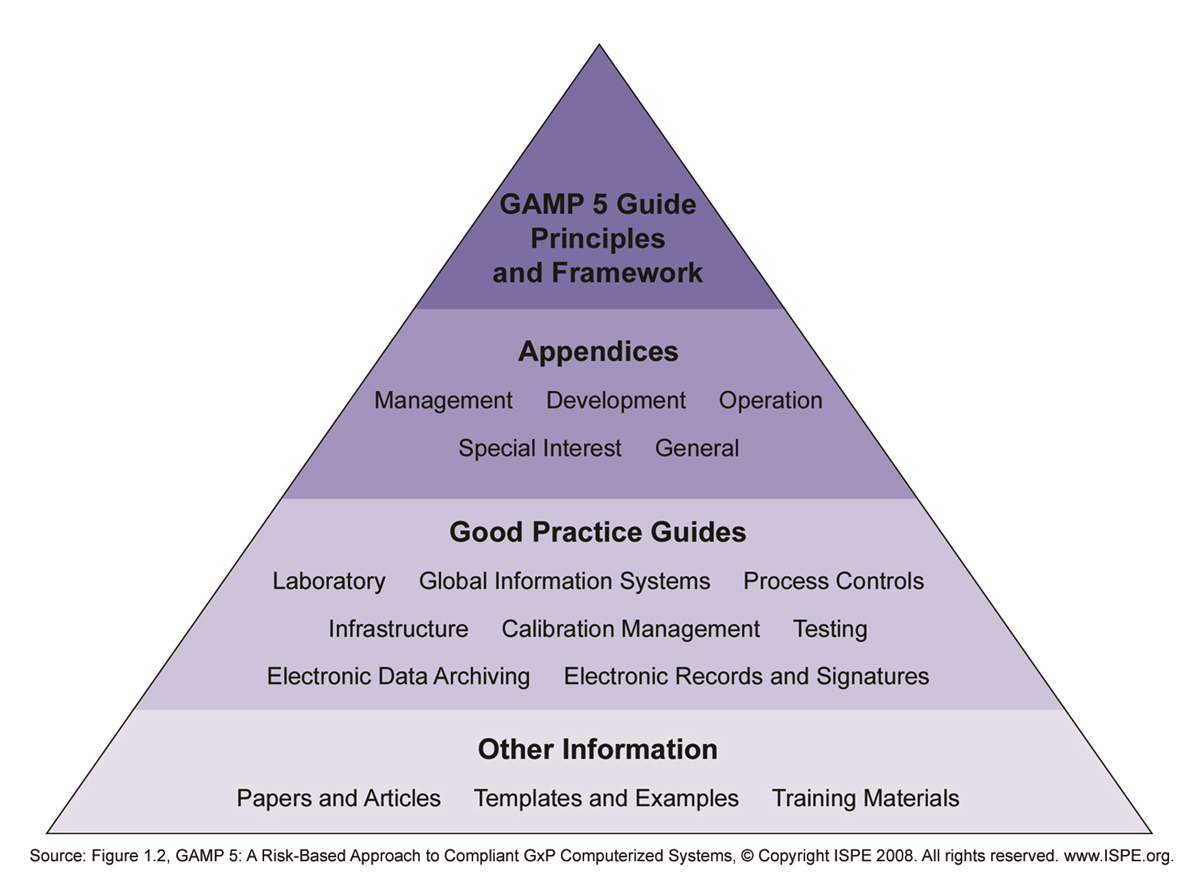
Validation

GAMP 5: A Risk-based Approach to Compliant Gxp Computerized Systems - Ispe Headquarters: 9781931879613 - AbeBooks
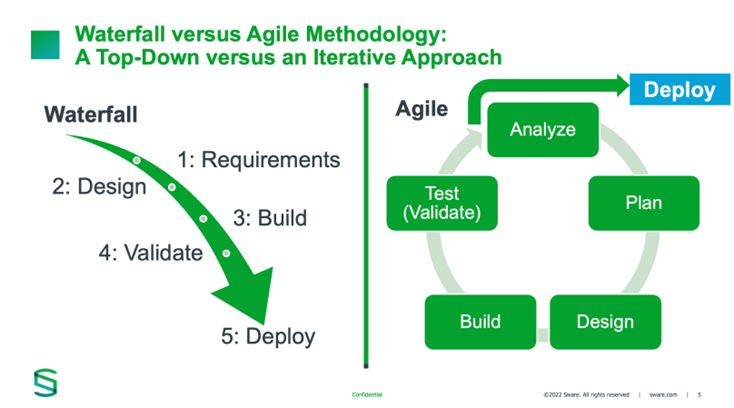
Advancing the Transition to Computer Software Assurance: Responding to the FDA Draft Guidance for Production and Quality System Software - Food and Drug Law Institute (FDLI)
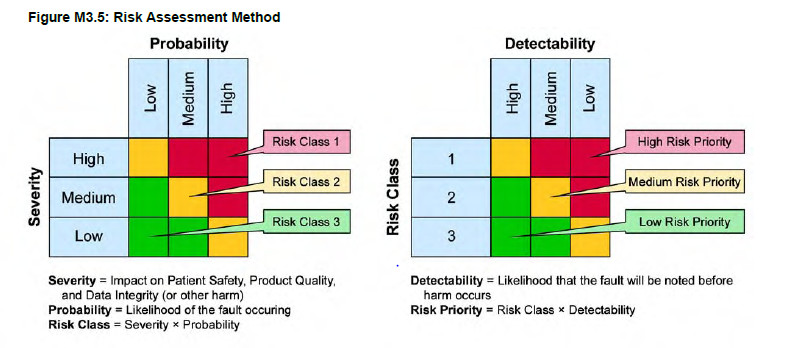
Risk Analysis Approach for Computerized Systems
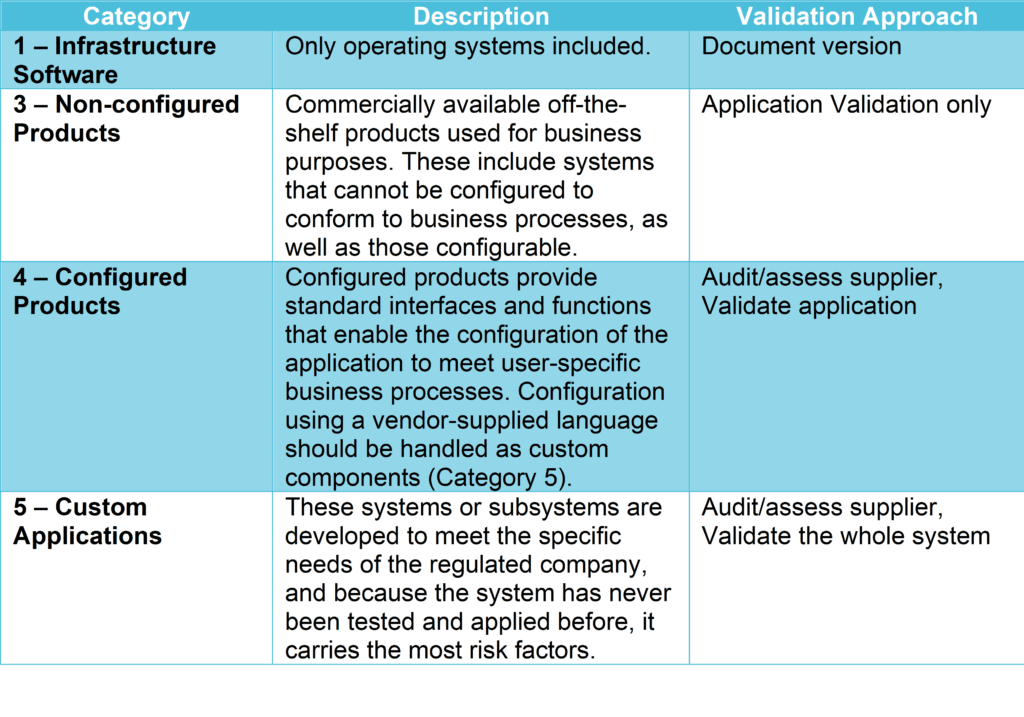
Risk-Based Computerized System Validation (CSV) and Computer Software Assurance (CSA) - Old Wine in a New Bottle? - Kvalito

GAMP 5: A Risk-based Approach to Compliant Gxp Computerized Systems

White Paper Gamp5 Risk Based Approach, PDF, Verification And Validation

GAMP Good Practice Guide: GxP Compliant Laboratory Computerized Systems 2nd Edition, ISPE

GAMP5 – Investigations of a Dog

GAMP® 5 Guide 2nd Edition Release
GAMP 5 for GxP Compliant Computerized Systems

ISPE GAMP 5 GUIDANCE SECOND - Issuu

Gamp 5 PDF